To get precise numbers use the test kit shown below. It is a Hach Total Hardness and Calcium test kit. Without such a precise and reliable test kit the calculations are impossible because tiny variations in the Calcium concentration greatly affect the overall final numbers. This is the test kit:
https://www.hach.com/hardness-total-calcium-test-kit-model-ha-4p/product?id=7640219511
Since GH is a sum of Ca + Mg we can measure GH and Ca and calculate the MG.
So the test kit gives us the values for GH and Ca an we determine Mg.
The aim is to get close to a ratio of Ca:Mg of 3-6:1.
Example with made up good numbers and the use of the German online calculator.
This example shows what combination of GH and Ca result in a good Ca:Mg ratio:
Go to the link below:
http://www.flowgrow.de/db/calculator/magnesium
Make sure that the radio button “Magnesium bestimmen” is selected.
First we will enter numbers that give us a good Ca:Mg ratio:
From experience I know that GH=7 and Ca=34 results in a good Ca:Mg ratio. So:
1. Enter 7 for “Gesamthärte (GH)”.
2. Enter 34 for “Referenzwert”Click the green button “Berechnen”.
The result is displayed in a white box to the right:
In this example we have 9.76 mg/L Mg (mg/L is equal to ppm). And the Ca:Mg ratio is 3.48:1.
Perfect.

Example with bad numbers – resulting in a Ca:Mg ratio that is not 3-6:1:
So you tested your water with the Hach test kit and got GH=4 and Ca=34 ppm. Looking at the example above you can already see that Ca=34 ppm should be paired with GH=7 to have a good Ca:Mg ratio. Meaning that in your water the Ca is too much for the GH of 4.
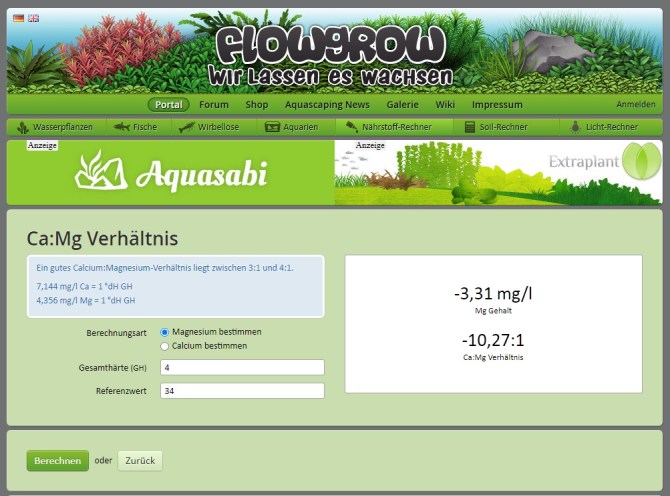
Now double check that using the German calculator:
1. Enter 4 for “Gesamthärte (GH)”.
2. Enter 34 for “Referenzwert”.
Click the green button “Berechnen”.
The result is displayed in a white box to the right:
You have -3.31mg/L Mg. And the Ca:Mg ratio is negative also.
That means that the Ca is way too much. So you need to increase the Mg.
::: How much Mg you need to add to your tank to get to the good ratio?
Look at the amount of Mg – it is -3.31. You want it to be 9.76 (like in the “Good Ca:Mg ratio” example. So you need to add enough MgCO3 (easily found on Ebay) to increase the Mg to 9.76.
You are starting from -3.31 and you need to get to 9.76. So you need to add about -3.31 + 9.76 = 13 ppm Mg.
::: So how much MgCO3 you need to add to an actual tank so you add 13 ppm of Mg?
We will use a 65 gallon tank as an example:
Go to this page:
http://www.flowgrow.de/db/calculator/dose
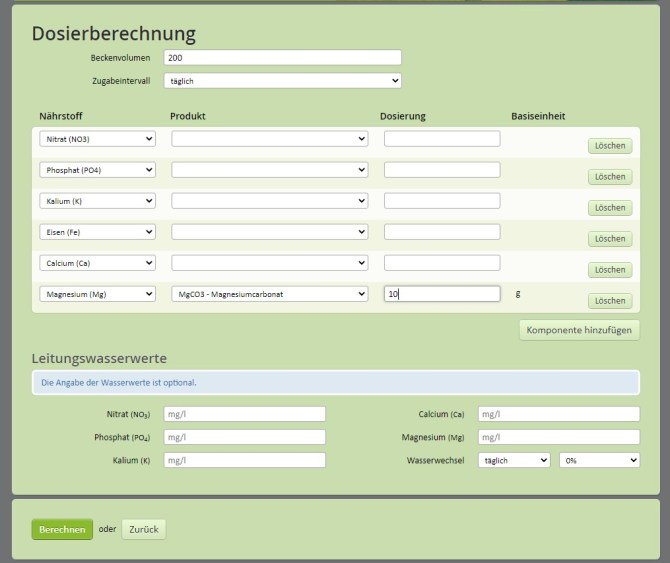
65 gallons is roughly 200 liters. Enter 200 in the “Beckenvolumen” box. It is approximate. From what I have seen 200 is a good approximation.
Find the drop box “Magnesium (Mg)” under “Nährstoff”. Leave it as it is.
In the drop box to the right of “”Magnesium (Mg)” choose “Magnesiumcarbonat MgCO3”.
Enter 10 in the box that has a “g” on the right. “g” is for “grams”.
Click the green button “Berechnen” (not “Loschen” which deletes this row of drop downs).
The results are shown UNDER the green button “Berechnen in a table named “Ergebnis”. So scroll down.
The table shows you how much of what element you added with your 10 grams of MgCO3. In this case it is 14.41 mg/L (=ppm).
For your tank: In your case you will enter your tank volume (we entered 200 liters) + grams of MgCO3 different from 10. Enter 10 and see if you get 14.41 as amount of Mg you added to your tank. Then change the 10 up and down until you get to 14.41. This will be the grams of MgCO3 for your actual tank.
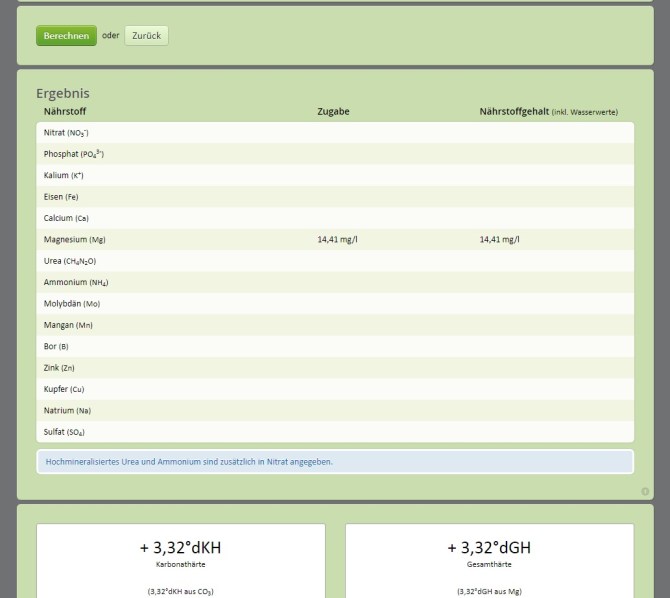
Now look BELOW that table and look at the two white boxes. They show how the KH and GH are affected by the addition of 10 g. of MgCO3. In this case KH was raised by 3.32 and also GH was raised by 3.32.
That means that we adjusted the GH and the Ca:Mg ratio should be good. But your starting KH of 4 may now read 7. That is not high but it is something to expect when we test next week. From my experience the KH will actiually jump only to about 6 max.
Important: Why not use Epsom Salt (MgSO4, Magnesium Sulphate)?
Note that other than Mg there are no extra chemical elements added. But if you use the common source of Mg – Epsom Salt – (“MgSO4.7H2O Bittersalz” in the German calculator) you will see that it adds Mg and Sulphur. There is no need for the Sulphur. If you add Epsom Salt to your tank to raise the Mg you are also constantly building the S. And never testing for it. Toxicity may happen and you don’t realize why. Same problem with fertilizers that contain Sodium and Chlorine. There are enough discussions and opinions how S, Na, and Cl are not harmful for a planted tank. They are all based on nothing. Better to just not add them if you can. That’s why you should use “clean” sources of fertilizer – like the MgCO3 or Dolomite (a natural mineral).
